As of January 1, 2021, the UK has continued to maintain many of the EU…


As of January 1, 2021, the UK has continued to maintain many of the EU…

CONTEXT: POST-BREXIT COVID-19 UK Taking into consideration the current sanitary situation in Europe and the…
An insight on the Medicines and Healthcare products Regulatory Agency Delivery Plan 2021-2023 and the…

Since the beginning of the pandemic of Covid-19, the usage of face mask has increased,…
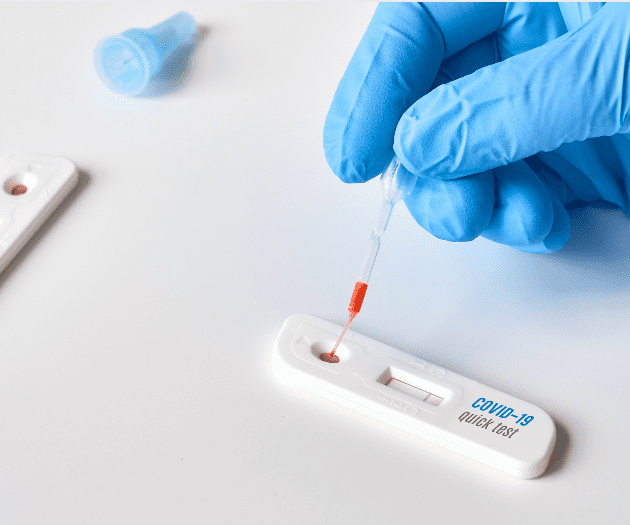
Due to the COVID-19 outbreak, the UK Medicines and Healthcare products Regulatory Agency (MHRA) has…

Following the Covid-19 crisis and the consequent surge in the demand of Covid test kits,…
Changes are changes, are they not? Brexit being a reality, it can now be presumed…

COVID-19, COVID-19… Needless to say, it is everyone’s duty to tackle and eliminate the virus…

The Medicines and Healthcare products Regulatory Agency (MHRA) is the standalone regulator for medicines and…

On 1 January, 2021, the United Kingdom left the EU Single Market, meaning that goods…