CONTEXT: POST-BREXIT COVID-19 UK
Taking into consideration the current sanitary situation in Europe and the world, the procurement of equipment to protect the population and health staff against the SARS-CoV-2 infection became a major concern in the UK as well. This turned out to be especially troublesome in the context of the UK’s withdrawal from the EU market, where the risk of shortages was patent.
To properly address this emergency, the British government and the MHRA agency on its behalf, published a set of guidelines on Regulatory status of equipment being used to help prevent coronavirus (COVID-19) .[/vc_column_text]
Timeline
The CE marking for medical devices will remain valid in the UK until 30 June 2023, as a part of the grace period agreed throughout the UK-EU Brexit negotiations. Special provisions apply to the case of Northern Ireland (NI), which has maintained a foot on the front door to the Union. NI continues to apply the EU law and will use the CE marking even after June 2023. However, a specific UKNI marking will also be required on the NI market.
COVID-19 PROTECTION EQUIPMENT IN THE UK
Today, the three most widely used items to fight against COVID-19 infection are hand sanitizer, masks, and gloves. Given that each of them comes in various models, having special properties, or reinforced action, procedures for authorities’ approval of those products in the UK may vary.
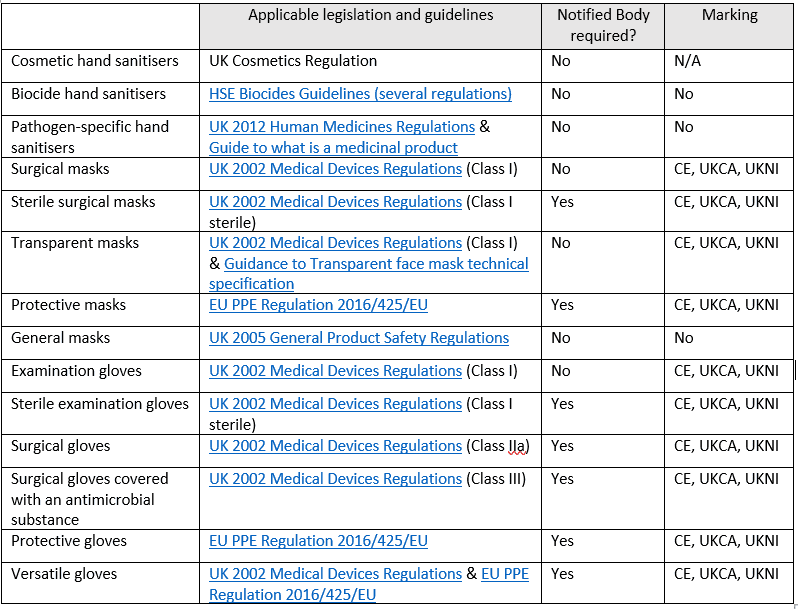
HAND SANITISERS
Hand sanitisers can be considered as either:
- COSMETICS: if primarily designed to clean or moisturise the skin and have some antimicrobial effectiveness. Soap is a typical example. The legislation currently applying to those products in the UK is the UK Cosmetics Regulation.
- BIOCIDES: if having active anti-microbial ingredients or with claims such as killing germs, disinfect or sanitise. The applicable legislation may come from different sources, and the MHRA recommends following the HSE guidance.
- MEDICINES: if claiming to fight against specific pathogens, such as coronaviruses. The applicable legislation, in this case, would be the Human Medicines Regulation 2012. You may also be interested in the MHRA’s Guide to what is a medicinal product, Appendix 5.
FACE MASKS
- MEDICAL DEVICES: all kinds of surgical, medical face masks are considered Class I medical devices or either, Class I(s), if supplied sterile. The 2002 UK Medical Device Regulation requirements apply in this case. Class I sterile devices require manufacturers to involve a Notified Body or the possession of a valid certificate. All surgical masks must wear the CE marking, UKNI or UKCA marking.
- In the scope of public interest, some surgical masks may be sold in the UK without wearing these markings, upon the approval of an exemption. Transparent face masks are also considered Class I medical devices but need to fulfill certain technical specifications.
- PERSONAL PROTECTIVE EQUIPMENT (PPE): refers to most masks designed to protect a person from a potential COVID infection. They are not considered as medical devices and, as such, they follow PPE Regulation 2016/425/EU. They need to display the CE or UKCA marking and must involve an EU Notified Body or UK Approved Body. Check here for the extended timeline to comply with UKCA.
- GENERAL PRODUCTS: or general face coverings that could be regarded as a cloth accessory. They do not wear any marking and a Notified/Approved Body Certificate is not needed. They follow the regulatory guidelines on the UK’s 2005 General Product Safety Regulations (GPSR). You can read more about transparent masks
GLOVES
- MEDICAL DEVICES: are all gloves to be used by health staff during surgery or patient examination to protect the patient. Examination gloves are, in general, considered as Class I medical devices, while surgical gloves are Class IIa devices under the 2002 Medical Devices Regulation. Sterile – Class Is and Class IIa gloves need an EU Notified Body or UK Approved Body’s certificate and must bear the CE, UKCA or UKNI marking. If surgical gloves are covered by an antimicrobial substance, they may be considered Class III devices (see Annex IX, Part II of the 2002 MDR). For more detailed guidelines on the medical devices conformity assessment follow this link.
- PERSONAL PROTECTIVE EQUIPMENT (PPE): gloves intended to protect the wearer from pathogen infection. They are regulated by the EU PPE Regulation (see the section related to masks). They must bear the CE, UKCA or UKNI marking and need an EU Notified Body or UK Approved ed Body’s certificate.
Some gloves could be used as both medical devices and PPE and must comply with the specific requirements of both regulations.
SUMMARY TABLE
COMPLIANCE
Manufacturers, UK responsible persons, importers and distributors must comply with the provisions of the applicable legislation. Practices such as unlawfully claiming to own a CE/UKCA/UKNI for medical devices or PPE may imply judicial prosecution by the relevant UK authorities.
Are you a COVID-19 test manufacturer placing your devices on the UK market? Contact us now and we will assist you in your path to compliance!
Alvaro Mercado Ortega
Regulatory Affairs Department
08.11.2021
Should you like to appoint Obelis UK as your UK RP, or if you simply need guidance on your UK documentation, please do not hesitate to contact us.
References
- Business companion. (2021). Cosmetics products. In-depth guide. Retrieved on 08/11/2021 from https://www.businesscompanion.info/en/quick-guides/product-safety/cosmetic-products
- Health and Safety Executive. Biocides regulation, supply and use. Retrieved on 08/11/2021 from Biocides regulation, supply and use – HSE
- Medicines and Healthcare products Regulatory Agency (2020). A guide to what is a medicinal product [PDF]. Retrieved on 08/11/2021 from pdf (publishing.service.gov.uk)
- Medicines and Healthcare products Regulatory Agency (2020). Medical devices: conformity assessment and the UKCA mark. Retrieved on 08/11/2021 from Medical devices: conformity assessment and the UKCA mark – GOV.UK (www.gov.uk)
- Medicines and Healthcare products Regulatory Agency (2020). Exemptions from Devices regulations during the coronavirus (COVID-19) outbreak. Retrieved on 08/11/2021 from Exemptions from Devices regulations during the coronavirus (COVID-19) outbreak – GOV.UK (www.gov.uk)
- Medicines and Healthcare products Regulatory Agency (2021). Regulatory status of equipment being used to help prevent coronavirus (COVID-19). Retrieved on 08/11/2021 from https://www.gov.uk/guidance/regulatory-status-of-equipment-being-used-to-help-prevent-coronavirus-covid-19
- Medicines and Healthcare products Regulatory Agency (2021). Transparent face mask technical specification. Retrieved on 08/11/2021 from Transparent face mask technical specification – GOV.UK (www.gov.uk)


